|
In this case, each sodium ion is surrounded by 4 chloride ions and each chloride ion is surrounded by 4 sodium ions and so on and so on, so that the result is a massive crystal. Arrange the solids in order of increasing melting points based on your classification, beginning with molecular solids.Ī. This is because sodium chloride ionic compounds form a gigantic lattice structure due to the electrostatic attractions between the individual ions.There are four types of crystal structures: Metallic. Based on their positions, predict whether each solid is ionic, molecular, covalent, or metallic. The easiest 3-D lattice to work with is the simple cubic lattice (SCC) which has lattice points on all the corners of a cube. These highly-ordered atoms form a structure known as a crystal lattice which extends in all 3D directions. Locate the component element(s) in the periodic table.Fracture mechanics was pioneered: the resistance of a structure could only be evaluated by considering its defects.\) as ionic, molecular, covalent, or metallic solids and arrange them in order of increasing melting points.Īsked for: classification and order of melting points The ionic and covalent bonds of ceramics are responsible for many unique properties of these materials, such as high hardness, high melting points, low thermal expansion, and good chemical resistance, but also for some undesirable characteristics, foremost being brittleness, which leads to fractures unless the material is toughened by. The experiments of Griffith put in evidence the effect of a defect as a scratch (which is actually a crack) on the material strength. For instance for metallic bonding, the attractive potential is due to the interactions between the negative electronic cloud and the positive nuclei, and is given by: (1.25) describes the formation of a vacancy and an interstitial in a silver lattice according to the KrögerVink notation. ionic crystal: A class of crystal consisting of a. Electrolytes Waldfried Plieth, in Electrochemistry for Materials Science, 2008 1.4.2 Intrinsic disorder Defects can be formed according to temperature and partial pressure. The expression of this potential is different for each kind of bonding but its shape remains similar. Inorganic solids often have simple crystal structures, and some of these structures are adopted by large families of ionic or covalent compounds. crystal lattice: A regular three-dimensional geometric arrangement of atoms, molecules, or ions in a crystal. Their arrangement varies depending on the ions sizes or the radius ratio (the ratio of the radii of the positive to the negative ion). form a crystal structure, there are certain rules or say guidelines which need. In this model each atom is supposed to have a certain radius r 0, determined by the equilibrium between the attractive and repulsive forces. Key Points Ions bound together by electrostatic attraction form ionic crystals. Most ceramic materials are neither purely covalently or ionically bonded. To model the bonding within the material, we consider the free electrons model. Covalent compounds share electrons between two or more bonded atoms whereas metallic compounds essentially share electrons with all the other metal atoms in the compound (a sea of electrons. 
This bonding is non-directional and will lead either to brittle or ductile (meaning experiencing plastic deformations before failure) materials as it will be explained.Īs previously stated, the nature of the bonding will have an effect on fracture behavior of the material. This leads to materials, such as metals and alloys, which are conductors. 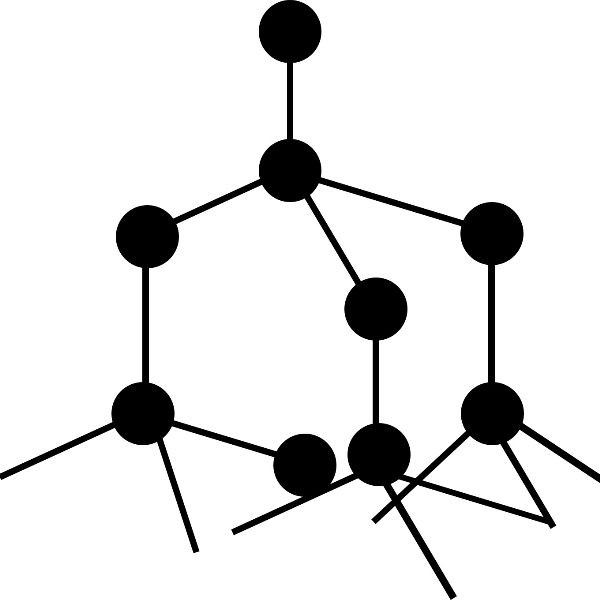 
Theses valence electrons are not shared between two atoms but are common to all the nuclei, and can thus move freely. Metallic bonding ( Picture I.12) is due to the existence of a cloud or sea of electrons. Covalent bonds are highly stable bonds with low melting. Picture I.12: Example of metallic bonding. Ionic compounds tend to be crystalline structures with high melting points that are water soluble.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
